GOOD STORAGE AND DISTRIBUTION PRACTICES FOR DRUG PRODUCTS

GOOD STORAGE AND DISTRIBUTION PRACTICES FOR DRUG PRODUCTS

Good manufacturing practices guide for drug products (GUI-0001

Good Distribution Practice for wholesale distribution of medicinal
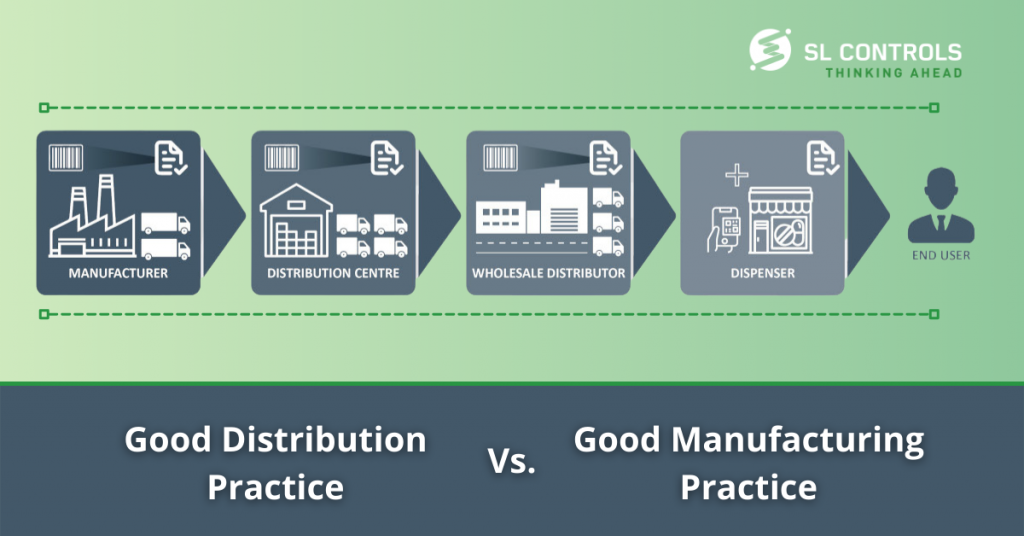
How Good Distribution Practice (GDP) differs from Good

PDF) Good Storage and Good Distribution Practices of

Quality Control During Drug Development

Annex 9 Guide to good storage practices for pharmaceuticals 1

Pharmaceutical Good Storage and Distribution Practices
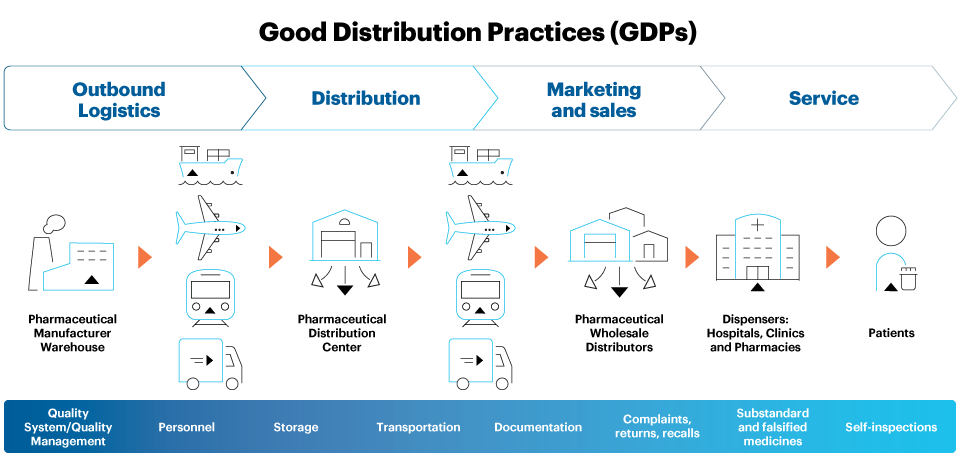
Good distribution practices require coordination, collaboration

GxP and cGxP in Bio/Pharmaceutical Industry - ppt download

Pharmaceutical Storage, Pharmaceutical Distribution

WHO, USP and PDA Make Substantial Contributions to GDP Guidance

The United States Pharmacopeia (USP) New Chapter on the Storage

Pharmaceutical Storage, Pharmaceutical Distribution

Good Storage Practices English, PDF


