FDA Clears Vagus Nerve Stimulator for Migraine Pain
FDA Clears Vagus Nerve Stimulator for Migraine Pain
The FDA has expanded the label for the gammaCore vagus nerve stimulator to include treatment of acute pain from migraine in addition to the previously cleared treatment of episodic cluster headache.

electroCore, Inc. on LinkedIn: What Is Cluster Headache? Symptoms, Causes, Diagnosis, and Treatment

GammaCore is FDA-Approved to Treat Migraine and Cluster Headache
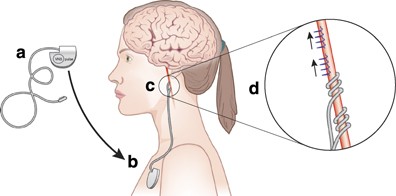
Noninvasive techniques for probing neurocircuitry and treating illness: vagus nerve stimulation (VNS), transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS)

FDA Greenlights New Wearable Device for the Treatment of Acute Migraine

Nerve Stimulation Devices for Migraine Treatment

New Device May End Pain for Veterans Suffering from PTSD and TBI-Related Headaches

Stimulating Your Vagus Nerve Can Help Relieve Migraine Pain - gammacore

FDA Clears Vagus Nerve Stimulator for Migraine Pain

Reviewing Noninvasive Vagus Nerve Stimulation for Migraine, Cluster Headache - Clinical Pain Advisor

Todd Charnetski on LinkedIn: This vagus nerve stimulation technology is amazing for headache prevention…

electroCore receives FDA Clearance for gammaCore (nVNS) for adjunctive use for the preventive treatment of Cluster Headache in adults