FDA Grants Fast Track to Palisade Bio's Drug Candidate

FDA Grants Fast Track to Palisade Bio's Drug Candidate
FDA Grants Fast Track to Palisade Bio’s Drug Candidate
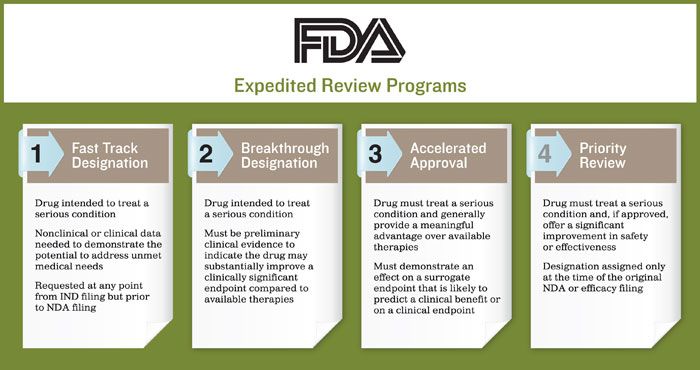
Accelerated Change: Understanding the FDA's Expedited Pathways
Insight Report: Where is the Peanut Allergy Drug Pipeline Now?

clbs-20221231

Production and clinical development of nanoparticles for gene delivery: Molecular Therapy Methods & Clinical Development

BBF11_Profiles_O-Z_P..
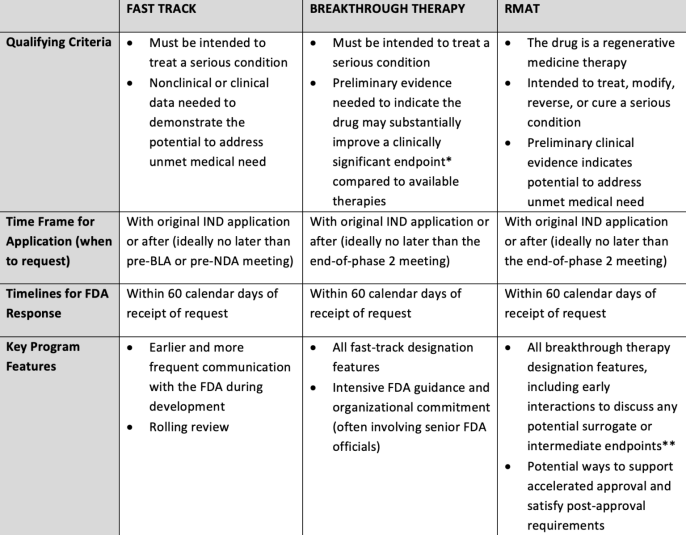
Overview of FDA Expedited Development and Approval Programs for Serious Conditions - SYNER-G

FDA's Expedited Development and Approval Programs

Clinical Trials Arena
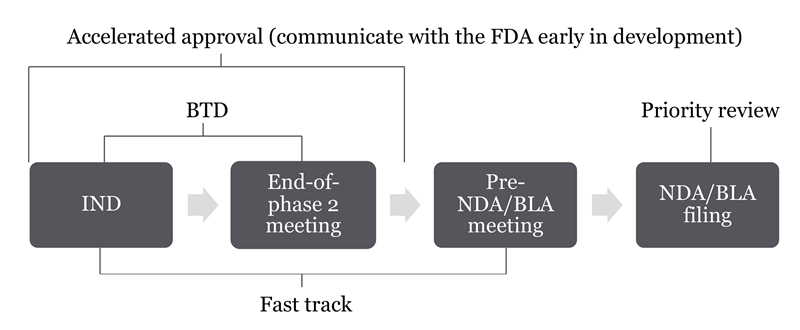
FDA Unveils Considerations for Rescinding Breakthrough Therapy Designation // Cooley // Global Law Firm
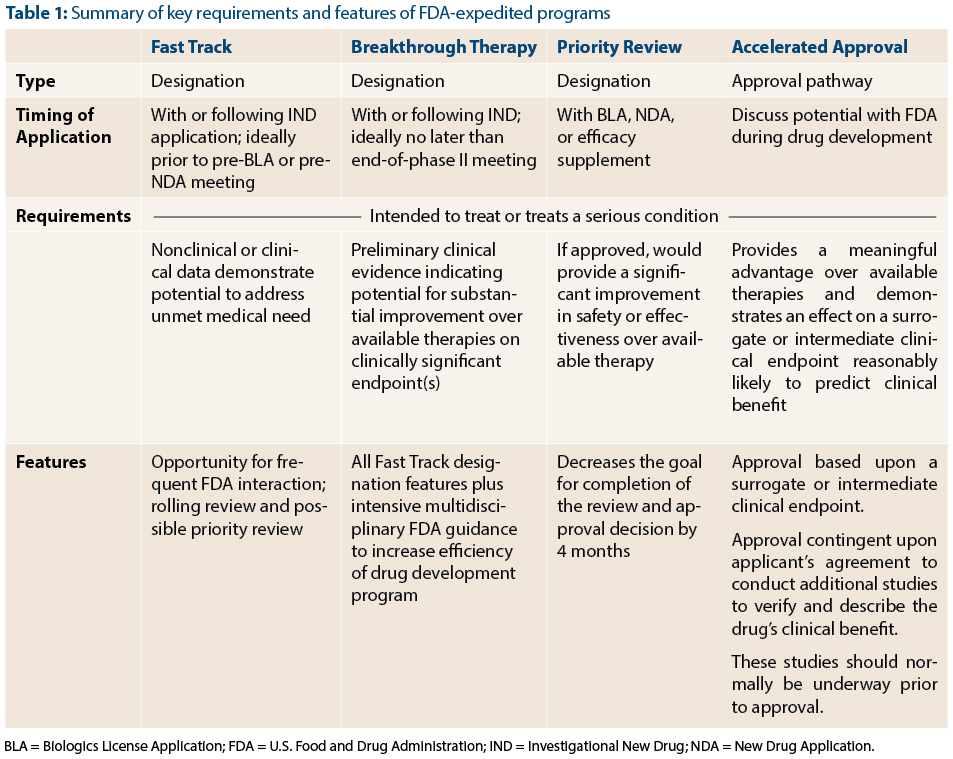
FDA Programs to Expedite Drug and Biologic Product Development - The ASCO Post
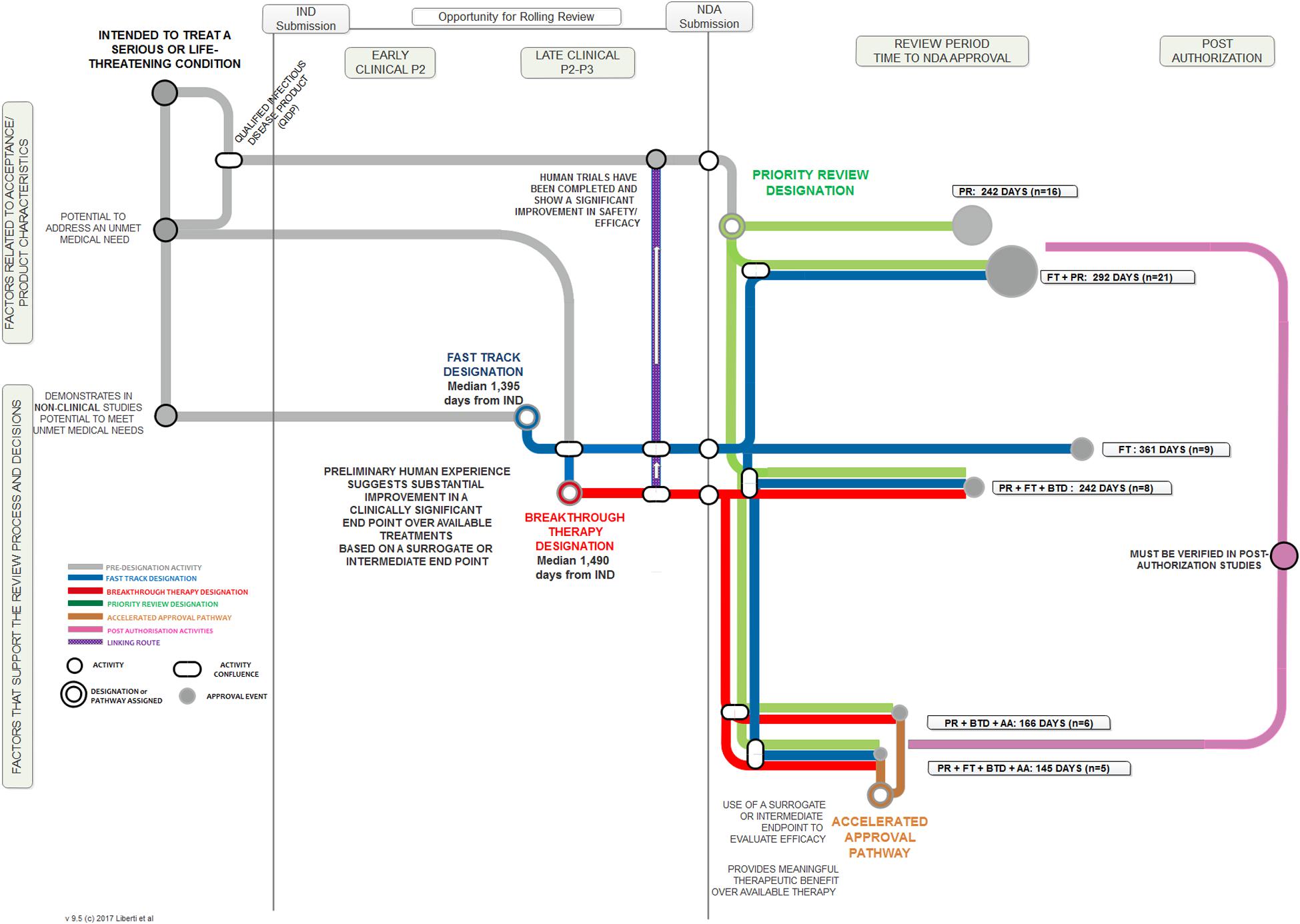
Frontiers FDA Facilitated Regulatory Pathways: Visualizing Their Characteristics, Development, and Authorization Timelines

Polymeric delivery systems for nucleic acid therapeutics: Approaching the clinic - ScienceDirect
Fast track (FDA) - Wikipedia
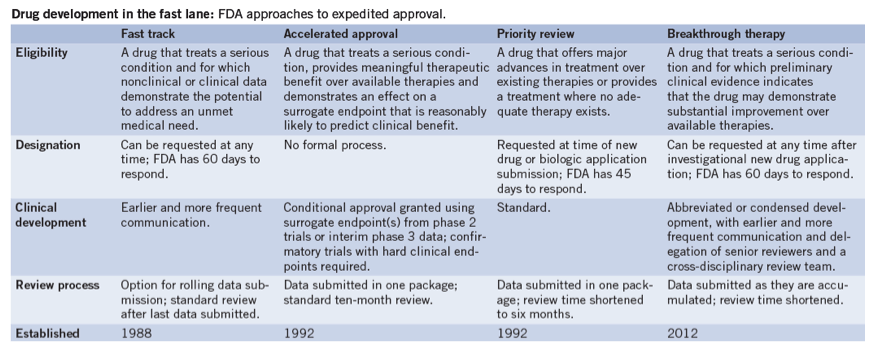
New FDA Approval Process: Breakthrough therapy

Palisade Bio to receive European patent for LB1148 - BioTuesdays


