Sky Medical Technology's geko NMES Device, USA
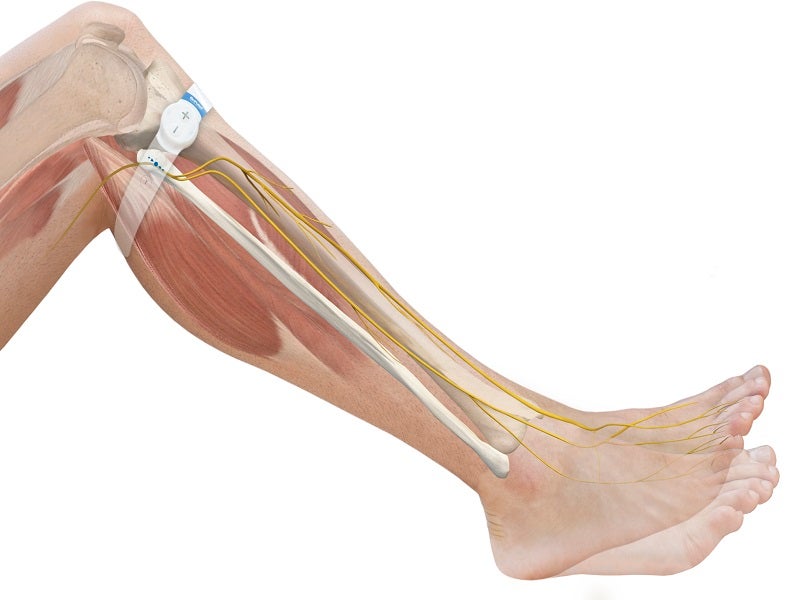
Sky Medical Technology's geko NMES Device, USA
The geko™ device is a neuromuscular electrostimulation (NMES) device designed to enhance blood flow to prevent venous thromboembolism (VTE).

PDF) The geko™ Electro-Stimulation Device for Venous Thromboembolism Prophylaxis: A NICE Medical Technology Guidance
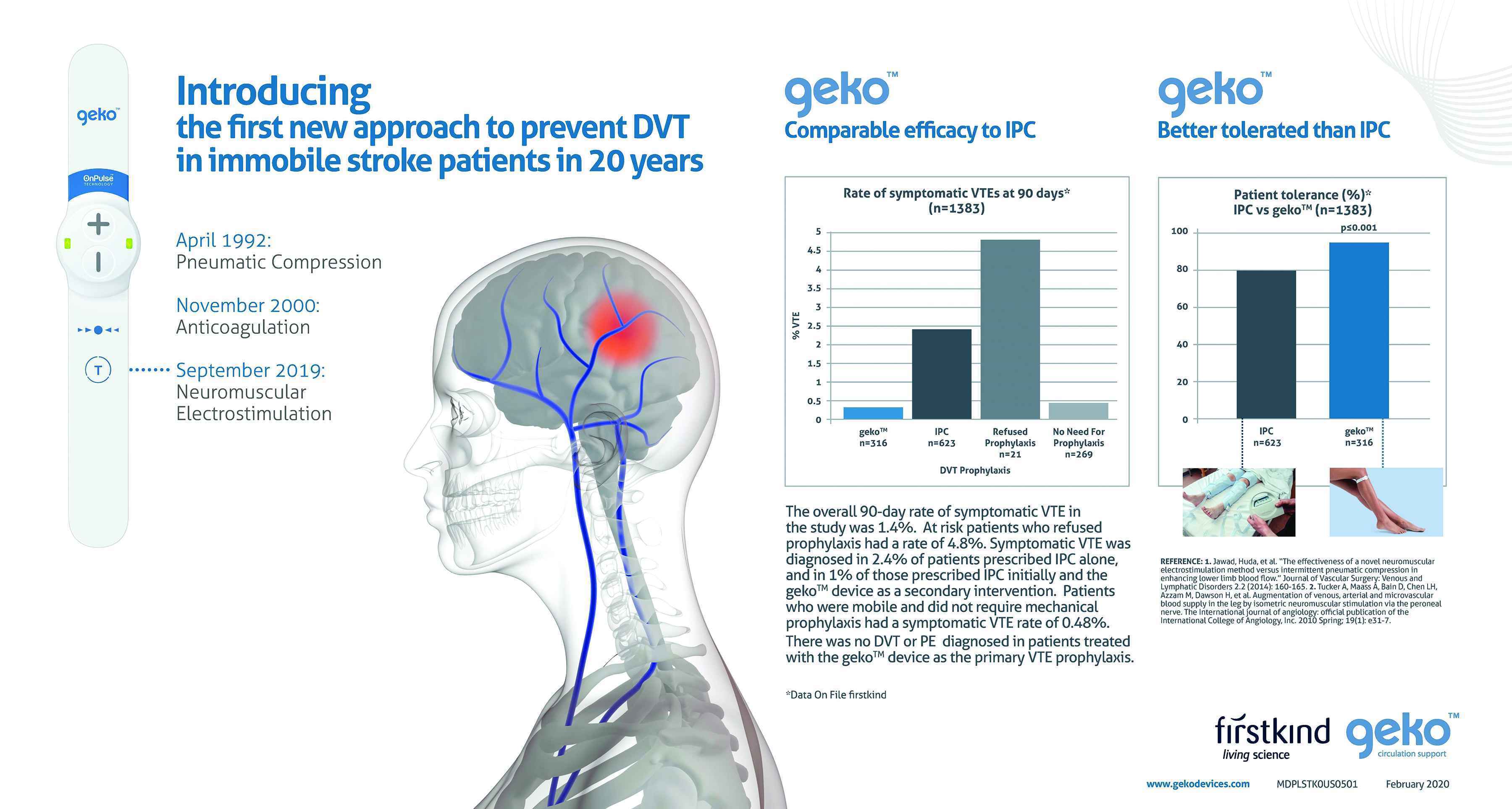
BullsEye Healthcare: 2020 - PharmaLive

The use of the geko™ device - Sponsored Symposium by Sky Medical

Sky Medical Technology Sky Medical Technology – part of DIT international showcase of UK Life Sciences innovators

Sky Medical Technology Bioelectronic medicine for clinicians

Mindray USA 121-001870-00 - McKesson Medical-Surgical

Sky wins FDA clearance to market the geko™ device for venous insufficiency and ischemia - a therapy area sorely in need of innovation.

Wound Masterclass - March 2023 by woundmasterclass - Issuu

The geko™ Electro-Stimulation Device for Venous Thromboembolism Prophylaxis: A NICE Medical Technology Guidance

FDA approves Sky Medical's geko device to prevent venous thromboembolism

The geko wound therapy device

The geko Device Makes its Debut at Major U.S. Orthopaedic Conference

The 2 Best Medical Alert Systems of 2024