FDA approves first-of-its-kind “living drug” for childhood

FDA approves first-of-its-kind “living drug” for childhood
The Food and Drug Administration on Wednesday approved a groundbreaking cancer treatment for childhood leukemia that uses patients' own genetically altered immune cells to fight the disease. It is the first gene therapy to be allowed in the United States - a "historic" approval, the agency said.

FDA Approves 'Absurd' Drug for Kids as Young as 8 to Delay Diabetes • Children's Health Defense
Rare Diseases: Considerations for the Development of Drugs and Biological Products Guidance Now Available - US FDA

Federal Register :: Authorizations of Emergency Use of Certain Drugs and Biological Products During the COVID-19 Pandemic; Availability

How the Pediatric Laws Work

Effectiveness of levofloxacin in the induction of chemotherapy in high-risk acute lymphoblastic leukaemia in children in a developing country - ecancer

FDA Approves First COVID-19 Treatment for Use in Children
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer_public/31/4e/314ea4df-859b-472e-994d-6bba73cefb9a/gettyimages-1156932872.jpg)
First Drug to Delay Type 1 Diabetes Approved by FDA, Smart News
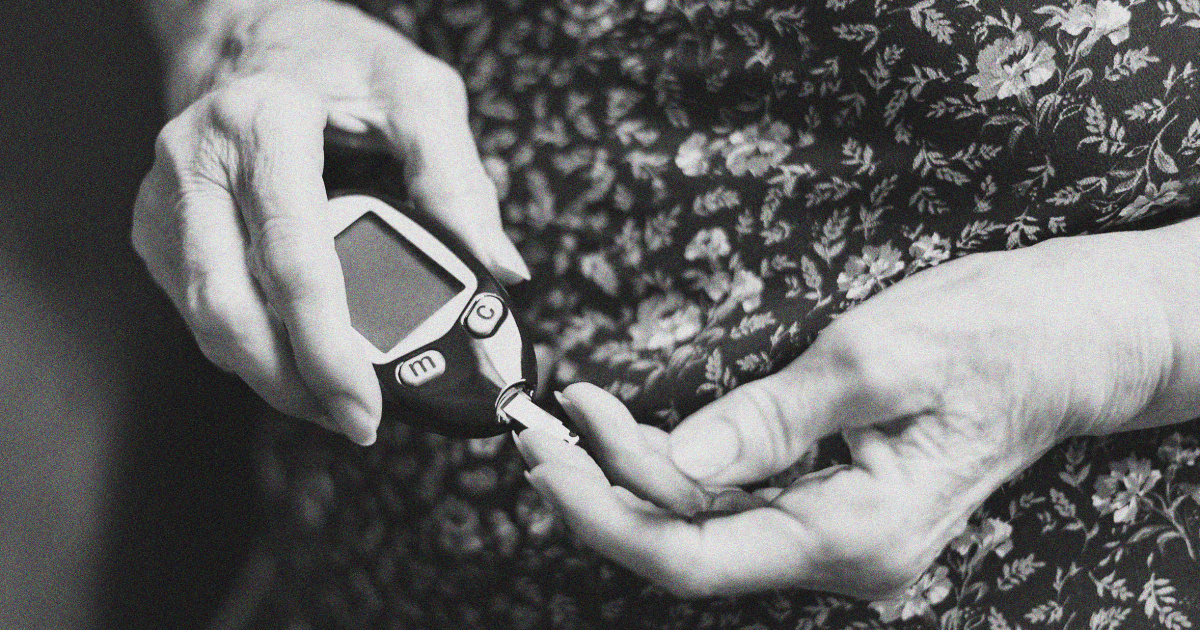
FDA approves 1st drug to delay the onset of Type 1 diabetes

FDA Approves First-of-Its-Kind Drug to Delay Type 1 Diabetes

New Drug Approval Moves Us Closer To Long-Term Control of Multiple Myeloma

FDA approves first-of-its-kind “living drug” for childhood leukemia – The Denver Post

FDA approves 1st drug to delay onset of Type 1 diabetes - ABC News

FDA approves semaglutide for obesity in adolescents
US Approves Drug to Slow Early Effects of Alzheimer's Disease


